Calculus for Kinetic Modeling
Velocity, Acceleration & Reaction Kinetics
Name___Sample Solution_____
Date___________
RK Exercise 3
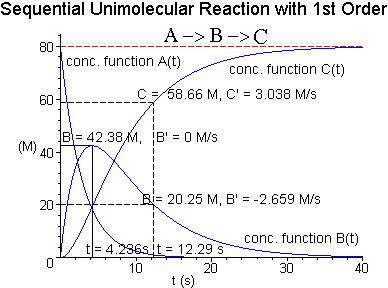
Reactant A has an initial concentration A(0)
= 80 M, where M = E-5 moles/liter. Let A->B->C be a sequential unimolecular
reaction with a rate constant of k1 = .35 for the first reaction, and a
rate constant of k2 = .15 for the second reaction.
(a) Compute velocity and acceleration functions
for the given concentration functions below.
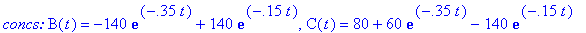
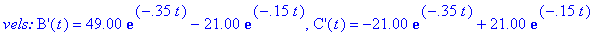
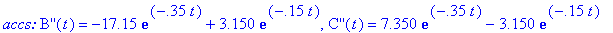 (b) How long after the reaction begins does it take
the concentration of intermediate B to reach a maximum? What is this maximum
concentration of intermediate B?
(b) How long after the reaction begins does it take
the concentration of intermediate B to reach a maximum? What is this maximum
concentration of intermediate B?
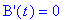
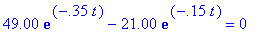
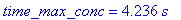
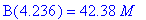 (c) When is the loss rate of B equal to -7/8 the
gain rate of C? What are these two rates?
(c) When is the loss rate of B equal to -7/8 the
gain rate of C? What are these two rates?
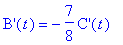
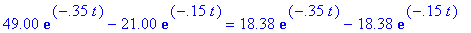
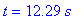
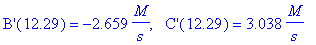 (d) What are the concentrations of intermediate B
and product C at the time found in (c)?
(d) What are the concentrations of intermediate B
and product C at the time found in (c)?
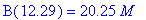 ,
, ![[Maple Math]](RK2ex3soln13.gif) (e) Label the concentration vs time plot below with
the information found in (b), (c), and (d).
(e) Label the concentration vs time plot below with
the information found in (b), (c), and (d).